Carney Complex
Clinical Definition
Carney complex is a rare dominantly heritable multiple endocrine neoplasia syndrome that affects endocrine glands including the adrenal cortex, the pituitary and the thyroid.
The syndrome is associated with many other nonendocrine tumors, including cardiac myxomas, testicular tumors, melanotic schwannoma, breast myxomatosis, and abnormal pigmentation or myxomas of the skin.
Carney complex has previously been known by the acronyms LAMB and NAME.
- LAMB is a mnemonic for the associated abnormalities (lentigines, atrial myxomas, and blue nevi) seen in patients with Carney complex syndrome.
- NAME is also a mnemonic for (nevi, atrial myxomas, and ephelides).
These syndromes are widely recognized to be main clinical manifestations of Carney complex.
The Carney complex, inherited in an autosomal dominant manner, is a highly penetrant disorder, and the diagnosis can be made at birth.
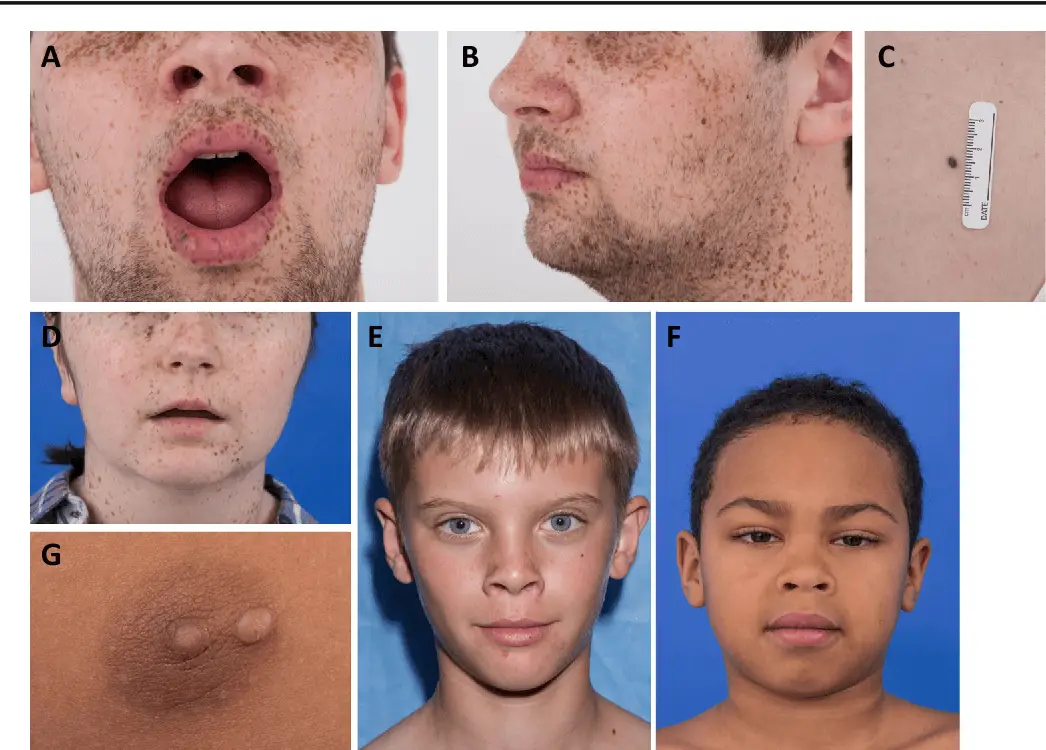 Figure X-1. Pigmentation in Carney complex; A and B: Lentigines on the vermilion border of the lips and the face; C: A blue nevus in a patient with the complex. D, E, and F: Other patients with Carney complex (and proven carriers of pathogenic PRKAR1A mutations) have variable pigmentation; some (like the patient in f) have very little and one has to look for other signs of the complex, such as a skin myxoma in the nipple area (g) | Illustration courtesy of Semantic Scholar Opens in new window
Figure X-1. Pigmentation in Carney complex; A and B: Lentigines on the vermilion border of the lips and the face; C: A blue nevus in a patient with the complex. D, E, and F: Other patients with Carney complex (and proven carriers of pathogenic PRKAR1A mutations) have variable pigmentation; some (like the patient in f) have very little and one has to look for other signs of the complex, such as a skin myxoma in the nipple area (g) | Illustration courtesy of Semantic Scholar Opens in new windowEndocrine abnormalities
- Cushing’s syndrome and PPNAD
Primary pigmented nodular adrenocortical disease (PPNAD) is found in more than 25% of patients with Carney complex and most frequently manifests as Cushing syndrome Opens in new window of hypercortisolism with truncal obesity, hypertension, edema, easy fatigability, hirsutism Opens in new window, and violaceous abdominal striae.
PPNAD Opens in new window is characterized by small-pigmented nodules, often surrounded by atrophic cortex. The disease is bilateral and is more frequent in females and in young adults, with a peak incidence during the 2nd and 3rd decades of life.
Diagnosis of PPNAD in a patient with Cushing syndrome is best made using Liddle’s test.
The administration of dexamethasone in the course of Liddle’s test establishes the diagnosis of PPNAD biochemically. Bilateral adrenalectomy is the most effective treatment.
- Pituitary tumors
Pituitary tumors in Carney complex typically develop from somatomammotrophs only and are often associated with gigantism and/or acromegaly; isolated prolactinomas are rare.
Acromegaly Opens in new window in Carney complex typically has a slow, progressive course.
Increased levels of IGF-1 and growth hormone can be present in up to 75% of Carney complex patients, whereas clinical acromegaly may be seen in only 10%–20% of the patients.
These biochemical abnormalities before the radiological evidence of a frank pituitary tumor are due to somatomammotrophic hyperplasia that precedes the development of tumors.
- Other endocrine tumors
Multiple thyroid nodules are present by ultrasonography in up to 75% of patients, Follicular adenoma is the most common finding; follicular or papillary thyroid cancer develops in less than 10% of patients.
Testicular tumors are a common finding in male patients with Carney complex and can be of three types:
- Large-cell calcifying Sertoli cell tumor— the most common testicular lesion in Carney complex appears in the first decade of life, often before puberty. It is almost always benign, with the exception of those in a few older patients who developed testicular cancer. And almost occurs bilaterally and is multifocal (in more than half of the patients). In addition, it presents as a calcification in ultrasonography.
- Testicular adrenal rest tumor — is due to (ectopic) PPNAD and can lead to Cushing’s syndrome.
- Leydig cell tumor — rare in Carney complex, can be seen alongside large-cell calcifying Sertoli cell tumors (unlike the latter, Leydig cell tumors are never seen isolated in the context of Carney complex).
Women with Carney complex commonly develop ovarian cysts Opens in new window and other tumors, including serous cystadenomas and teratomas. These tumors may progress occasionally to ovarian carcinoma (mucinus adenocarcinoma or endometrioid carcinoma); reported cases of ovarian cancer occurred in the fifth decade of life.
Nonendocrine abnormalities
- Cardiac myxomas
Cardiac myxomas Opens in new window are neoplasms found relatively frequently among patients with Carney complex (30%–40% of all Carney complex patients develop at least one cardiac myxoma in their lifetime).
Although benign by nature, these tumors cause most of the mortality among patients with Carney complex because of their location.
Cardiac myxomas may be within any chamber of the heart, often at multiple sites, and tend to recur frequently. Once they appear, they tend to grow rather fast, sometimes within the course of weeks.
Cardiac myxomas can grow in utero, during infancy, and recur even in the elderly; in other words, screening for these tumors should be undertaken at any age, although most tumors are seen in young adults.
Early detection of cardiac myxomas by echocardiography and their surgical excision are essential to prevent death or severe morbidity due to embolism, strokes, or cardiac failure.
- Skin lesions
Skin lesions are among the major criteria of Carney complex. The most common skin lesions are lentigines, epitheliod blue nevi, and cutaneous myxomas.
Other skin lesions, such as Spitz nevi Opens in new window, café-au-lait spots Opens in new window, and hypopigmented macules have all been reported but they are rarer than the ones mentioned above.
- Breast lesions
Breast tissue lesions in Carney complex include lobular or nodular myxomatosis, myxoid fibroadenomas, or ductal adenomas.
Despite extensive involvement of the breast in Carney complex, there does not appear to be increased risk for breast cancer in this condition.
- Psammomatous melanotic schwannomas
Psammomatous melanotic schwannomas Opens in new window are present in 8% of Carney complex. These lesion can be found anywhere in the peripheral nervous system, but are most frequently found in the gastrointestinal tract (oesophagus and stomach) and the paraspinal sympathetic chain.
Malignancy may be observed in 10% of the cases, with frequent metastasis to the lungs, the liver, the brain, and/or the spinal cord.
- Bone lesions
Osteochondromyxoma Opens in new window is a bone tumor found in less than 5% of patients with Carney complex. It occurs early in life, usually before the age of 2, and clinically presents as a painless mass in distal long bones (diaphyseal) and small flat bones (nasal).
- Cancers
In addition to thyroid, ovarian, and testicular carcinomas, hepatocellular and rare pancreatic tumors (acinar cell carcinoma, adenocarcinoma, and intraductal pancreatic mucinous neoplasm) were recently described among Carney complex patients.
Mestastatic adrenocortical carcinoma has also developed in at least two patients with a background of PPNAD.
Clinical Manifestations and Diagnosis
Carney complex can present with a number of endocrine and non-endocrine manifestations which vary between patients
Genetics of Carney complex
Genetic linkage analysis identified two independent chromosomal loci for Caney complex: 17p22–4 and 2p16. The gene on 2p16 remains unknown.
In most cases, Carney complex is caused by inactivating mutations in PRKAR1A, which is located on 17q22–4 and encodes the most widely expressed of the regulatory subunits of protein kinase A, type 1 alpha.
Diagnostic criteria
The diagnosis of Carney complex is made if two or more major manifestations of the syndrome are present (see Table X-1).
The diagnosis may also be made when only one of the major criteria is present and the patient is a carrier of a known inactivating mutation of PRKAR1A (see Supplemental criteria, Table X-1).
| Table X-1 | Diagnostic criteria for Carney complex |
|---|
Major diagnostic crieteria
Supplemental criteria
|
| Abbreviations: GH, growth hormone; LCCSCT, large-cell calcifiying Sertoli cell tumor; PPNAD, pigmented nodular adrenocortical disease. Reproduced from Horvath A, Stratakis CA, Carney complex and lentiginosis, Pigment Cell & Melonoma Research, Volume 22, Issue 5, pp. 580-587, Copyright (2009) with permission from John Wiley and Sons. |
Therapeutics
The goal of management of patients with Carney complex is the early detection of clinically important manifestations at a presymptomatic stage through regular clinical and biochemical screening.
Pediatric patients
The following is recommended for pediatric patients with Carney complex:
- echocardiographic evaluation should be performed during the first 6 months of life and annually thereafter; if there is a history of an excised myxoma, the evaluation should be performed every 6 months
- other imaging or hormonal screening in prepubertal children is not considered necessary, unless there are clinical issues, such as, for example, growth failure or overgrowth; obesity; or precocoious puberty.
Post-pubertal children and adult patients
In post-pubertal children and adult patients, the following annual workup is recommended:
- an echocardiogram
- UFC (diurnal cortisol levels and an overnight 1 mg dexamethasone testing may be performed as needed and if clinical symptoms of Cushing’s syndrome exist)
- serum IGF-1 levels
- an oral glucose tolerance test, to measure levels of growth hormone
- a testicular ultrasound (if calcification suggesting the presence of a large-cell calcifiying Sertoli cell tumor was seen during the initial workup)
- a thyroid ultrasound should be obtained at the initial evaluation and then repeated annually as needed; fine-needle aspiration should be performed if large nodules are present
- an ovarian ultrasound is recommended at the initial evaluation and repeated if an abnormality was detected
- brain and spine MRIs should be obtained during the initial evaluation of any adult patient with Carney complex, but not repeated, unless clinical neurological signs suggest the possibility of a schwannoma (psammomatous melanotic schwannoma)
- Anselmo J. Medeiros S, Carneiro V, et al. A large family with Carney complex caused by the S147G PRKAR1A mutation shows a unique spectrum of disease including adrenocortical cancer. J Clin Endocrinal Metab 2012; 97: 351-9.
- Bartherat J. Horvah A, Groussin L, et al. Mutations in regulatory subunit type 1A of cyclic adenosine 5’-monophosphate-dependent protein kinase (PRKAR1A): Phenotype analysis in 353 patients and 80 different genotypes. J Clin Endocrinal Metab 2009;94: 2085-91.
- Briassoulis G, Kuburovic V, Xekouki P, et al. Recurrent left atrial myxomas in Carney complex: A genetic cause of multiple strokes that can be prevented. J Stroke Cerebrovasc Dis 2012; 21:914e1-e8.
- Carney JA and Toorkey BC. Myxoid fibroadenoma and allied conditions (myxomatosis) of the breast. A heritable disorder with special associations including cardiac and cutaneous myxomas. Am J Surg Pathol 1991;15:713-21.
- Carney JA, Boccon-Gibod L, Jarka DE, et al. Osteochondromyxoma of bone: A congenital tumor associated with lentigines and other unusual disorders. Am J Surg Pathol 2001;15:164-76.
- Chrousos GP and Stratakis CA. Carney complex and the familial lentiginosis syndromes: Link to inherited neoplasias and developmental disorders, and genetic loci. J Intern Med 1998;243:573-9.
- Gennari M, Stratakis CA, Hovarth A, et al. A novel PRKAR1A mutation associated with hepatocellular carcinoma in a young patient and a variable Carney complex phenotype in affected subjects in older generations. Clin Endocrinol (Oxf) 2008; 69:751-5.
- Kirschner LS, Carney JA, Pack SD, et al. Mutations of the gene encoding the protein kinase A type I-alpha regulatory subunit in patients with the Carney complex. Nat Genet 2000; 26:89-92.
- Pringle DR, Yin Z, Lee AA, et al. Thyroid-specific ablation of the Carney complex gene, PRKAR1A, results in hyperthyroidism and follicular thyroid cancer. Endocr Relat Cancer 2012; 19:435-46.

